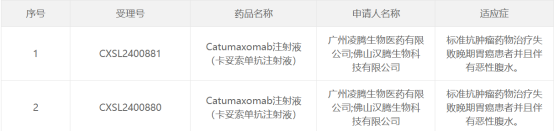
Recently, Canton Biologics announced a significant advancement in its collaboration with Guangzhou Lingten Biomedicine Co., Ltd. (“Lingten Bio”). The domestic Phase III clinical IND for Catumaxomab injection – a bispecific antibody transferred from Germany to Canton Biologics by Lingten Bio’s strategic partner Lindis Biotech – has been officially approved by China’s National Medical Products Administration (NMPA). The proposed indication is for the treatment of late‑stage gastric cancer patients with malignant ascites who have failed standard antitumor therapy.
Local Process Breakthrough to Accelerate Access to Innovative Drugs
Catumaxomab, the first bispecific antibody approved worldwide, received marketing authorization from the European Medicines Agency (EMA) in 2009. As Lindis Biotech’s partner in China and Asia, Lingten Bio transferred the manufacturing process of Catumaxomab injection from overseas to China and entrusted Canton Biologics with completing the process transfer and the production of drug substance and drug product for Phase III clinical samples.
Leveraging years of accumulated experience in bispecific antibody process development and external technology transfer, as well as manufacturing quality standards compliant with NMPA, EMA, and FDA requirements, Canton Biologics successfully completed the cross‑border transfer of the manufacturing process and analytical methods, the production of Phase III clinical drug substance and drug product, stability studies, and characterization studies – all key elements of the comparability exercise. The comparison results demonstrated that the product quality and safety of the Phase III clinical samples manufactured at Canton Biologics’ facility were highly consistent with those of the original European process.
The approval of Lingten Bio’s Phase III IND for Catumaxomab not only validates Canton Biologics’ capabilities in cross‑border technology transfer and international‑standard manufacturing and quality assurance for large‑molecule biologics CDMO services, but also highlights the strategic value of local suppliers in ensuring supply chain stability and reducing production costs. This milestone lays a solid foundation for Canton Biologics to provide commercial manufacturing services for large‑molecule drugs to more domestic and international clients.
Addressing Clinical Challenges and Filling Unmet Medical Needs
Malignant ascites is one of the most common and serious complications in patients with advanced cancer. Traditional treatment options have limited efficacy and are associated with high recurrence rates. By targeting both EpCAM and CD3 through its bispecific mechanism of action, Catumaxomab injection activates the patient’s immune system to precisely eliminate malignant ascites lesions, potentially overcoming the limitations of existing therapies and offering patients a more effective and safer treatment option.
Following this Phase III IND approval, Lingten Bio will conduct multi‑center clinical studies in collaboration with several top medical institutions in China, and Canton Biologics will continue to provide high‑quality drug manufacturing support.

 中文
中文